Peptide Publications Archive
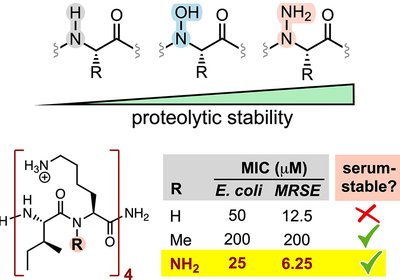
Backbone Heteroatom Shield
Del Valle Lab
Replacing backbone amide nitrogens with nitrogen or oxygen heteroatoms can slow protease degradation without sacrificing the folded structures peptides need to work. A tetra-N-aminated antimicrobial …
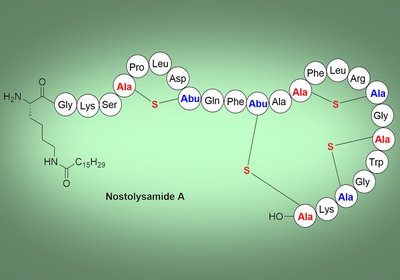
Breaching Candida
van der Donk Lab
A class II lanthipeptide from the cyanobacterium Nostoc punctiforme kills Candida species by disrupting cell membranes, marking the first antifungal activity reported for this lanthipeptide …
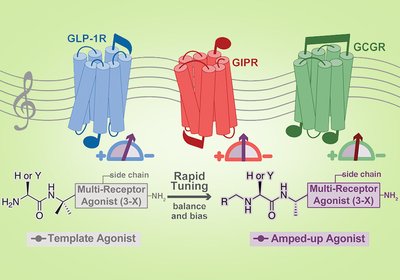
Tuning Incretin Balance
Kumar Lab
A single chemical edit at the N-terminus of a GLP-1/GIP/glucagon multiagonist can shift receptor potency balance by orders of magnitude, selectively suppress β-arrestin recruitment, or …
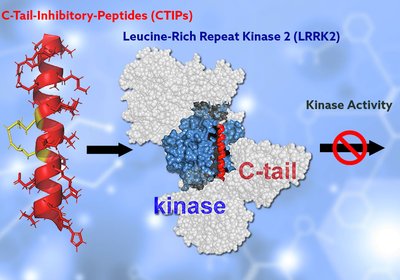
Stapling LRRK2
Kennedy Lab
Hydrocarbon-stapled peptides that mimic the C-terminal helix of LRRK2 can penetrate cells, bind the kinase directly, and suppress its hyperactivation without triggering the mislocalization side …
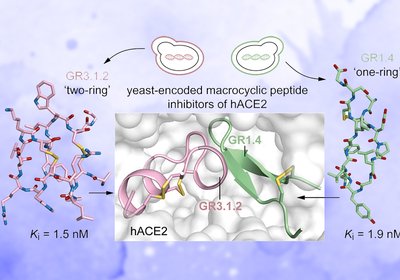
Yeast Finds Inhibitors
Angelini Lab
Yeast display technology, long used to evolve antibodies, can now rapidly surface macrocyclic peptide inhibitors of human angiotensin-converting enzyme 2 with potencies in the low-nanomolar …
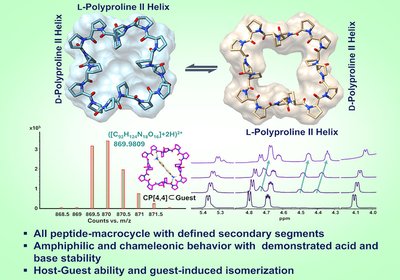
Chameleonic Proline Rings
Palma Lab
A new family of all-peptide macrocycles built from alternating L- and D-proline tetramers flips between two distinct conformations depending on solvent, solves long-standing functionalization challenges …
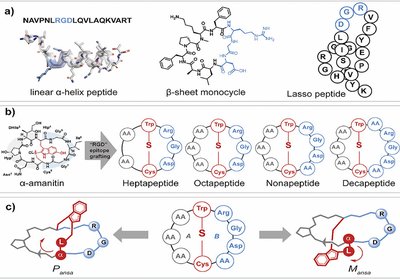
Ansamers Target Integrins
Yao Lab
Bicyclic peptides derived from the amanitin scaffold can exist as non-interconvertible conformational isomers called ansamers, and the two mirror-like forms can differ by orders of …
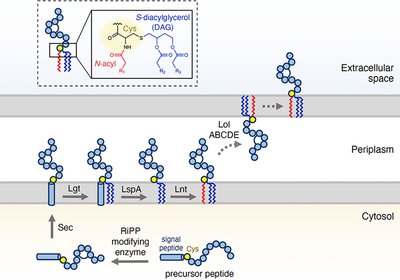
Lipid-Hijacking RiPPs
van der Donk Lab
Bacteria already possess a well-oiled machinery for anchoring lipoproteins to their membranes. A new study reveals that a family of ribosomally synthesized peptides has co-opted …
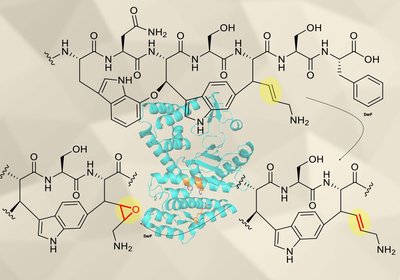
DarF Decoded
Phan Lab
An enzyme in the darobactin biosynthetic pathway had been classified as a protease for years. New biochemical evidence shows it is actually an α-ketoglutarate-dependent oxidase …

Porous Collagen Crystals
Merg Lab
Collagen-mimetic peptides are renowned for triple-helix rigidity, but no one had channeled that rigidity into a porous crystalline framework. Now, by appending a single alkyl …
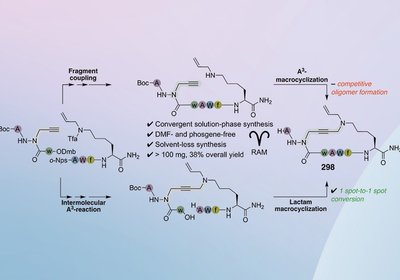
Scalable Azapeptide Macrocycles
Lubell Lab
A convergent solution-phase strategy for a potent macrocyclic azapeptide CD36 modulator sidesteps preparative chromatography entirely, delivering 100 mg of >99%-pure material by swapping an oligomer-prone …
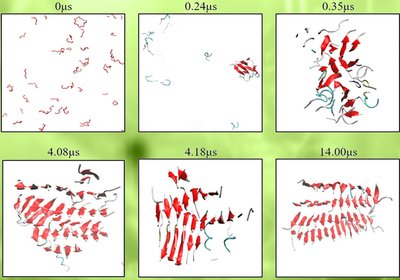
Hairpins Hold the Key
Hall, Radford, and Brockwell Labs
A single methionine swap in α-synuclein's N-terminal P1 motif dismantles the β-hairpin architecture that normally launches amyloid fibril formation
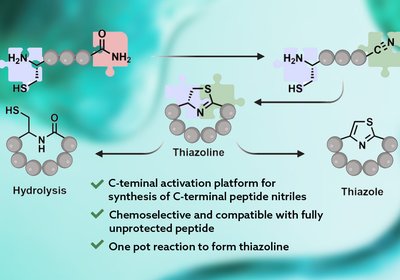
Activating Native Termini
Raj Lab
Thiazoline and thiazole macrocycles embedded in bioactive marine peptides have long required pre-built specialty building blocks that slow synthesis and risk epimerization. A new palladium-mediated …
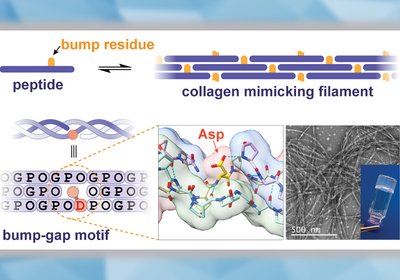
Bump-Gap Filaments
Chenoweth Lab
Natural collagen carries risks of immunogenicity and resists chemical modification, but synthetic collagen-mimetic peptides have struggled to match its continuous, mechanically robust fibrillar architecture. A …

Labeling Serine Selectively
Chen Lab
Serine and threonine hydroxyl groups are among the most difficult side chains to modify selectively in complex peptides, yet they govern phosphorylation, glycosylation, and enzyme …
Chaperone Orientation Rules
Gierasch Lab
Three closely related Hsp70 molecular chaperones grip the same model peptides in strikingly different orientations: bacterial DnaK shows no directional preference, human Hsc70 leans toward …
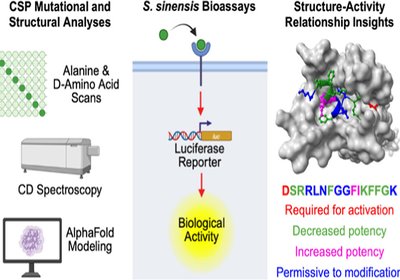
Silencing Strep Signals
Tal-Gan Lab
Streptococcus sinensis is an oral commensal that turns deadly when it reaches the bloodstream, causing infective endocarditis with roughly 25% mortality. A new mutational map …
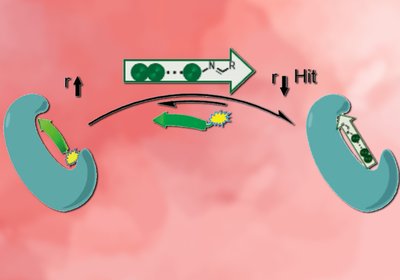
Fragment Capping PDZ
Wilson Lab
β-strand-mediated protein–protein interactions are among the hardest targets in chemical biology, and PDZ domains are notoriously resistant to small-molecule ligands. A dynamic ligation screen against …
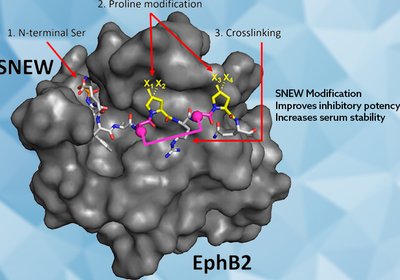
Stabilizing SNEW
Sawyer Lab
The EphB2 receptor drives several cancers, yet its best peptide inhibitor, SNEW, has languished with modest potency and poor serum stability. Swapping its N-terminal serine …
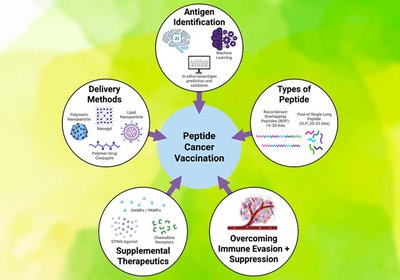
Priming the Attack
Kimmel Lab
No peptide cancer vaccine has yet earned FDA approval, yet the field is converging on strategies that may finally close that gap. A new review …
Hunting Huntingtin
Harding Lab
Huntingtin, the protein at the center of Huntington's disease, has long resisted selective chemical interrogation. A new suite of macrocyclic peptide tools now binds it …
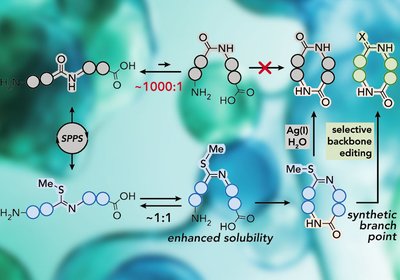
Unlocking Small Macrocycles
VanVeller Lab
Short homochiral peptides routinely defeat macrocyclization attempts, either failing to close the ring or dissolving poorly enough to prevent even trying. A single backbone thioimidate …
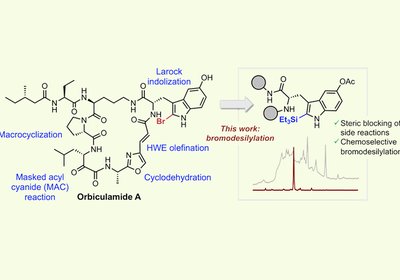
Taming Bromine
Perrin Lab
For 35 years, the marine macrocycle orbiculamide A resisted total synthesis, largely because its 2-bromo-5-hydroxytryptophan residue defeats every conventional bromination strategy. Now, a chemoselective late-stage …
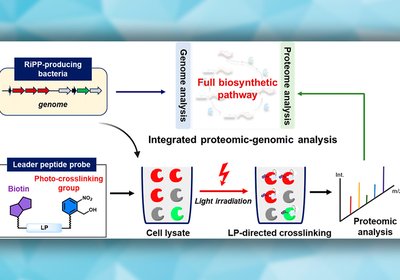
Mapping Hidden Enzymes
Wang Lab
Genome mining reliably maps biosynthetic gene clusters for ribosomally synthesized and post-translationally modified peptides, RiPPs, yet routinely misses enzymes encoded elsewhere in the genome. A …