Peptide Publications Archive
Showing items in Synthesis & Methodology · Clear filter
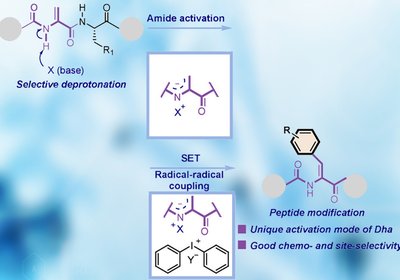
Activating Dehydroalanine
Liu and Wang Groups
A previously overlooked deprotonation of the dehydroalanine amide N–H unlocks selective arylation of peptides under mild conditions, with no metal catalyst required
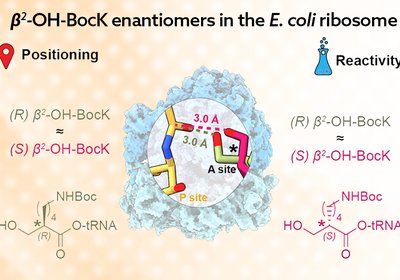
Backbone Sensitivity
Cate & Schepartz Groups
Researchers in the Cate and Schepartz Groups at the University of California, Berkeley, and the Lawrence Berkeley National Laboratory, published in the Journal of the …
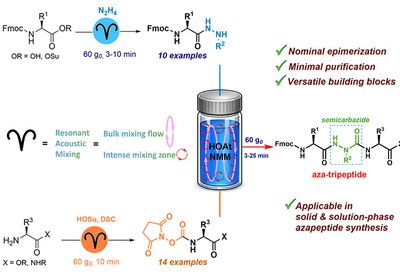
Scaling Azapeptides
Lubell Lab
A resonant acoustic mixing strategy solves long-standing problems in azapeptide synthesis, delivering scalable aza-tripeptide building blocks without epimerization, hydantoin formation, or chromatographic purification
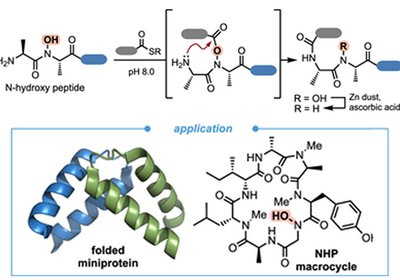
Backbone Bonds Unlocked
Del Valle Lab
A backbone hydroxamate group enables sequence-agnostic peptide ligation in water, bypassing thiolated amino acids and opening convergent routes to folded miniproteins and macrocyclic natural products
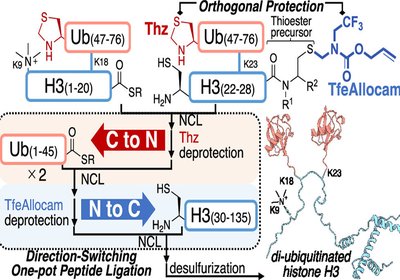
Switching Ligation Directions
Hayashi Lab
Researchers in the Hayashi, Murakami, and Okamoto Groups at Nagoya University and the University of Tokyo, published in the Journal of the American Chemical Society …

Templated Macrocycles
Wennemers Group
Researchers in the Wennemers Group at ETH Zürich, published in Science, developed a bifunctional tripeptide catalyst that templates head-to-tail macrocyclization through dual substrate engagement
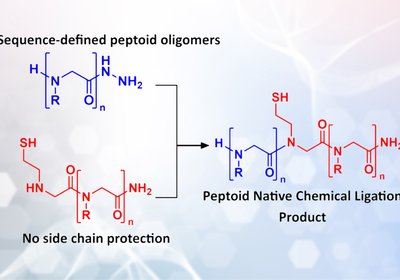
Peptoid Ligation
Kirshenbaum Group
Researchers adapt native chemical ligation to peptoid oligomers, enabling convergent synthesis of sequence-defined macromolecules and functional nanomaterials
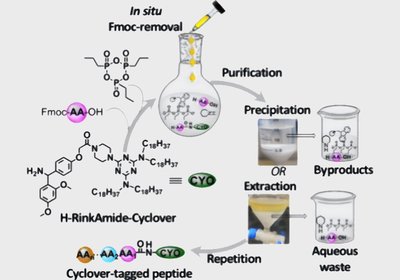
Greener Peptide Couplings
Albericio & de la Torre Group
Researchers in the Albericio & de la Torre Group at the University of KwaZulu-Natal, published in Organic Letters, introduced Cyclover as a commercially accessible …
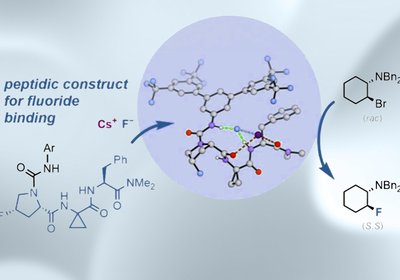
Peptides Deliver Fluoride
Gouverneur Lab
Fluorine is among the most consequential substituents in medicinal chemistry, improving metabolic stability, membrane permeability, and target binding across a broad range of drug candidates
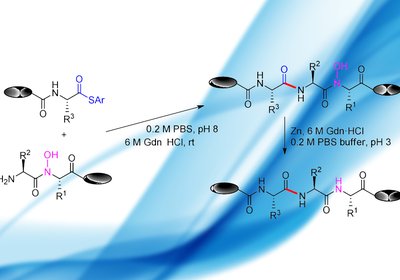
Site-Free Ligation
Hua Fu Lab
Researchers in the Fu Group at Tsinghua University in Beijing, published in Org. Lett., have developed a new chemoselective peptide ligation strategy, hydroxylamine-involved ligation, …
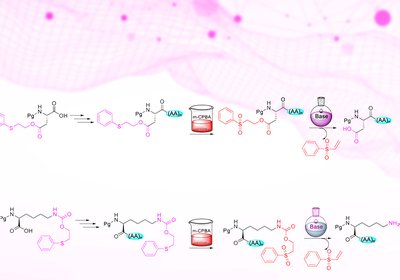
Safety Catch Strategy
Albericio Lab
Researchers supervised by Professor Fernando Albericio at the University of KwaZulu-Natal developed a new family of safety-catch protecting groups based on the phenylthioethyl, Pte, and …
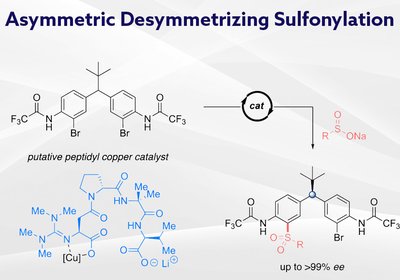
Remote Stereocontrol
Miller Lab
Researchers in the Miller Group at Yale University, published in the Journal of the American Chemical Society, have developed guanidinylated peptide ligands that enable …
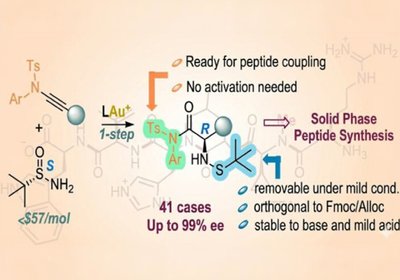
Ready-Made Amino Acids
Liming Zhang Lab
Researchers in the Zhang Group at the University of California, Santa Barbara, published in the Journal of the American Chemical Society, developed a concise …

Green Peptide Synthesis
Wellings, Meldal, and Wade Groups
Researchers supervised by Dr. Donald A. Wellings, Nobel Laureate Professor Morten Meldal, and Professor John D. Wade at the University of Melbourne, published in Nature …
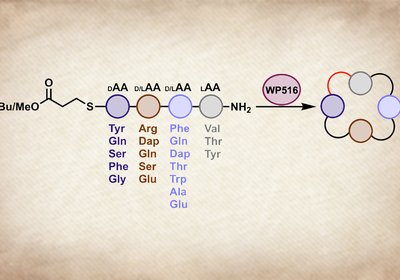
Tetrapeptide Cyclase
Parkinson Lab
Cyclic tetrapeptides occupy a tantalizing corner of chemical space. Head-to-tail macrocyclization eliminates the charged termini that limit membrane permeability, while the constrained
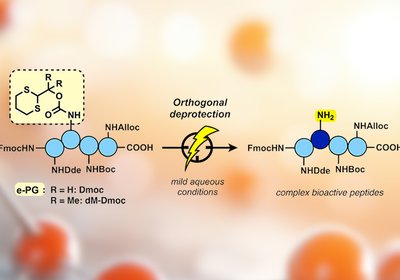
Electrochemical Deprotection
Malins Lab
Protecting group chemistry sits at the heart of peptide synthesis. Every amide bond formed on a growing chain demands that other reactive side chains stay …
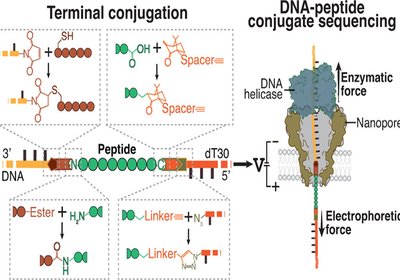
Threading Peptide Sequences
Dekker & Albada Labs
Researchers in the Dekker Group at Delft University of Technology, in collaboration with Bauke Albada at Wageningen University, published in the Journal of the American …
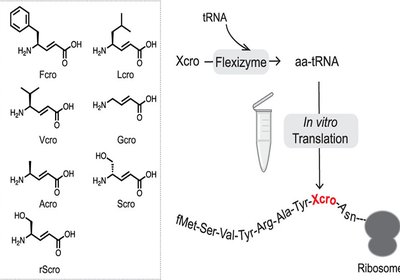
Ribosomal γ-Amino Acids
Suga Lab
Researchers in the Hiroaki Suga Group at the University of Tokyo hypothesize in the Journal of the American Chemical Society, that the conformational constraint …
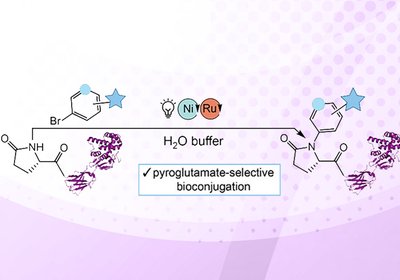
Pyroglutamate Labeling
Ball Lab
Scientists in the laboratories of Professors Zachary Ball and Laura Segatori at Rice University have developed a photoredox catalysis method that enables site-selective modification of …
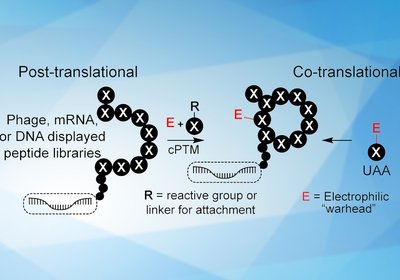
Covalent Library Design
Derda Group
Researchers in the Derda Group at the University of Alberta, published in Biochemistry, have surveyed the emerging field of covalent genetically-encoded libraries for discovering …
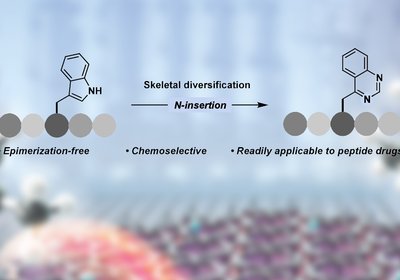
Tryptophan Transformation
Grob and Morandi Labs
A collaboration between the laboratories of Professor Bill Morandi in the Laboratorium für Organische Chemie and Dr. Nathalie Grob in the Institute of Pharmaceutical Sciences, …
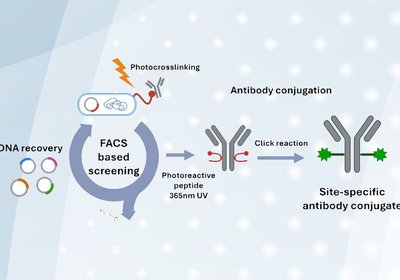
Evolved Photo-Cross-Linkers
Yoo & Kim Labs
Antibody conjugates represent a cornerstone of modern therapeutics, diagnostics, and imaging. By attaching payloads such as cytotoxins, fluorophores, or radionuclides to immunoglobulins, researchers can harness …
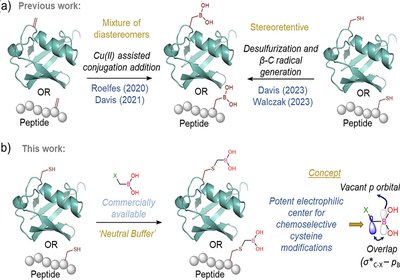
Cysteine Boronation
Bandyopadhyay Lab
Boronic acids have earned a versatile reputation in chemical biology and drug design. Their empty p orbital allows dynamic switching between sp2 and sp …
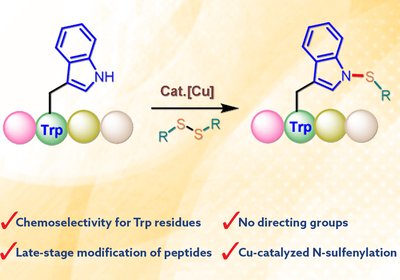
Tryptophan N-Sulfenylation
Yuan Lab
Peptides have become increasingly important as drug candidates and research tools due to their favorable safety profiles and ability to modulate protein-protein interactions. However, native …