O-GlcNAcylation is a dynamic intracellular posttranslational modification, PTM, in which the monosaccharide β-N-acetylglucosamine, GlcNAc, is attached to serine or threonine residues by O-GlcNAc transferase, OGT, and removed by O-GlcNAc hydrolase, OGA. Because UDP-GlcNAc is synthesized via the hexosamine biosynthetic pathway, which integrates carbohydrate, amino acid, and fatty acid metabolism, cellular O-GlcNAc levels serve as a real-time readout of metabolic state. Dysregulation of this modification is implicated in cancer, neurodegeneration, and other human diseases, yet its site-specific biochemical consequences remain difficult to disentangle using classical cell-based approaches alone.
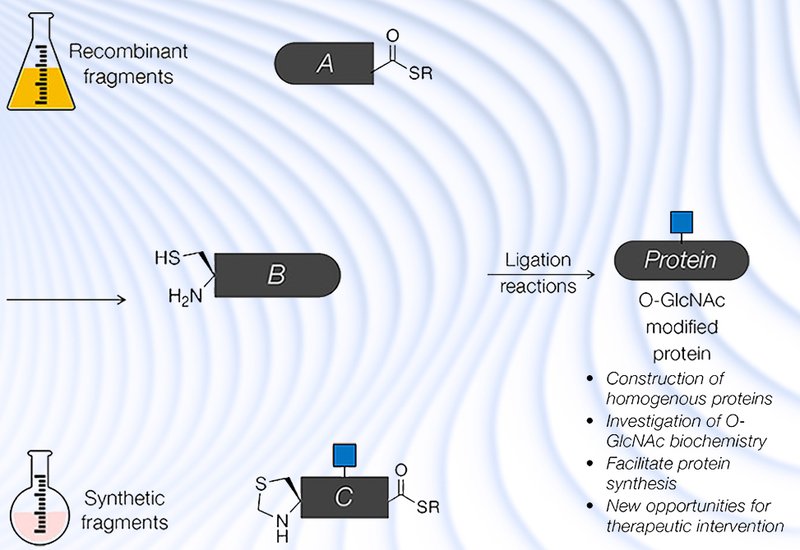
Researchers in the Pratt Lab at the University of Southern California, published in Biochemistry, perspective the chemical and enzymatic strategies that make site-homogeneous O-GlcNAc-modified proteins accessible, then survey how those tools have illuminated OGT and OGA mechanism, reshaped understanding of disease-relevant proteins, and opened new territory in glycoprotein synthesis. The perspective draws heavily on work from the authors' own group alongside contributions from collaborators and the broader field.
On the mechanistic front, synthetic glycopeptide libraries and structural biology have begun to map OGT substrate recognition. Crystal structures reveal that peptide substrates bind in an extended conformation within a cleft between the tetratricopeptide repeat and catalytic domains, and kinetic studies point to an ordered sequential bi-bi mechanism. For OGA, structures of the enzyme in complex with glycopeptides derived from αB-crystallin, TAB1, ELK1, and Lamin B1 show that substrates bind bidirectionally within an arm-in-arm homodimeric cleft, with extensive hydrogen-bonding to the GlcNAc moiety driving recognition. A separate substrate-specificity study demonstrated that OGA displays stringent selectivity for the native sugar, in sharp contrast to OGT's broad donor tolerance.
The most striking disease-relevant findings concern α-synuclein and HSP27. O-GlcNAcylation of α-synuclein at S87, installed via expressed protein ligation, EPL, forces the protein into amyloid fibrils with a distinct cryo-EM-resolved core architecture and unique inter-protofilament salt bridges. Although aggregation kinetics in vitro are similar to those of the unmodified protein, preformed fibrils of the gS87 proteoform show markedly diminished seeding activity, reduced Lewy body formation, and attenuated pathology in primary neurons and mouse models. For HSP27, O-GlcNAcylation at T184 enhances chaperone activity by disrupting an autoinhibitory IXI/ACD domain interaction, promotes protein refolding through the BAG3/HSP70 co-chaperone system, and significantly delays caspase-3 cleavage by caspase-9, connecting a single PTM to both proteostasis and apoptosis regulation. In the realm of PTM cross-talk, semisynthetic Akt1 bearing gS473 partially activates the kinase toward GSK3 peptide substrates, a result that contradicts the prevailing assumption that O-GlcNAcylation at this site is purely inhibitory.
The perspective also highlights O-GlcNAc as a practical synthetic tool. The removable glycosylation modification, RGM, strategy exploits transient O-GlcNAcylation to stabilize folding intermediates during chemical synthesis of disulfide-rich proteins, enabling the first total chemical synthesis of the interleukin-5 homodimer with correctly formed intermolecular disulfides. A complementary lysine-masked GlcNAc, LMG, strategy developed by Chen et al. improves the solubility of aggregation-prone glycopeptides during solid-phase peptide synthesis, SPPS, and can be removed under standard native chemical ligation, NCL, conditions to reveal native glycosylation that is then elaborated into diverse glycoforms.
Taken together, these advances position protein semisynthesis as the method of choice for decoding site-specific O-GlcNAc biology. The authors argue that OGA inhibitors already in clinical development for tauopathies may warrant evaluation in Parkinson's disease, supported by the α-synuclein fibril-strain data. Broader adoption will require cheaper access to O-GlcNAc-modified amino acid building blocks, longer SPPS-accessible peptide lengths, and more robust desulfurization and deselenization protocols, challenges the field is actively addressing.