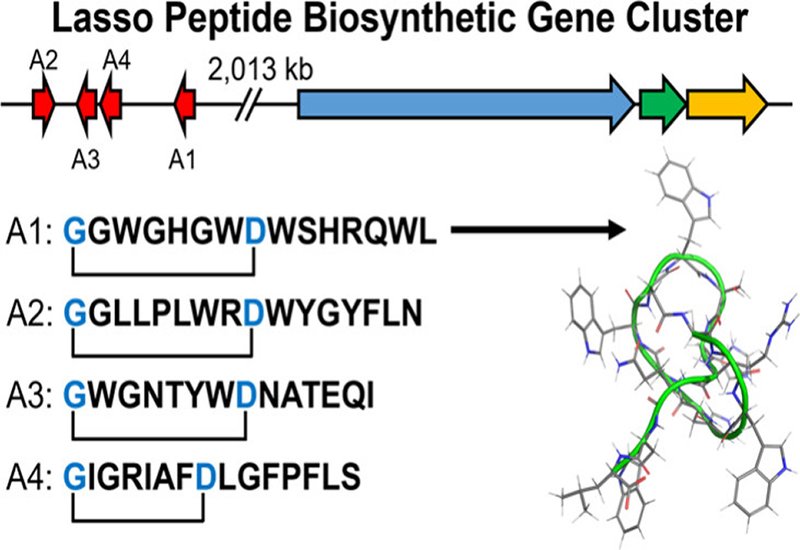
Lasso peptides are ribosomally synthesized and post-translationally modified peptides, RiPPs, distinguished by their threaded, knotted topology. A macrolactam ring forms between the N-terminus and an aspartate or glutamate side chain roughly seven to eight residues downstream, and the C-terminal tail threads through this ring to create a molecular lasso. This unique architecture has made lasso peptides a focal point for biosynthetic studies, and all examples characterized to date share a common genetic blueprint: the precursor peptide gene sits adjacent to the modification enzymes that process it. This colocalization ensures coordinated expression of substrate and enzymes. Most lasso peptides also target bacteria, typically by inhibiting RNA polymerase. The cebulassopins challenge both of these trends.
Researchers in the Seyedsayamdost Group at Princeton University and Moon Group at Kyung Hee University, Korea, discovered four new lasso peptides from the marine actinomycete Saccharopolyspora cebuensis SPE 10-1 and reported the findings in the Journal of the American Chemical Society. The team cultured the bacterium to stationary phase and screened the metabolome using ultra-performance liquid chromatography coupled to quadrupole time-of-flight mass spectrometry, UPLC-qTOF-MS. Two peaks with molecular formulas consistent with large peptides caught their attention. They isolated the compounds and solved their structures using one-dimensional and two-dimensional NMR spectroscopy, high-resolution tandem mass spectrometry, HR-MS/MS, and advanced Marfey's analysis to confirm L-configuration of all amino acid residues. Cebulassopin A contains 15 residues with an eight-residue macrolactam and a seven-residue tail, while cebulassopin B contains 16 residues with a nine-residue ring and seven-residue tail. Nuclear Overhauser effect spectroscopy constraints confirmed the lasso topology, and computational modeling with XPLOR-NIH revealed steric locks provided by histidine and tryptophan residues in cebulassopin A and by tyrosine residues in cebulassopin B.
Whole-genome sequencing of the producing strain revealed the genetic surprise. AntiSMASH analysis identified three lasso peptide biosynthetic gene clusters, BGCs, but only one matched the cebulassopin sequences. This cluster, named cbp, encodes a macrolactam synthetase, CbpC, a RiPP recognition element, CbpB1, and a protease, CbpB2. However, it lacks any precursor peptide genes in the immediate vicinity. A manual search located four short open reading frames, cbpA1 through cbpA4, encoding precursor peptides that perfectly matched cebulassopins A and B. These precursor genes sit more than 2 million base pairs away from the biosynthetic enzymes, a completely unprecedented arrangement for lasso peptides or any other RiPP class. The researchers validated this split architecture through isothermal titration calorimetry, demonstrating tight binding between the precursor CbpA1 and the recognition element CbpB1 with a dissociation constant of 1.5 micromolar. Comparative genomics revealed 16 additional cebulassopin-like precursors in public databases, eight from split BGCs in related S. cebuensis strains and eight from intact BGCs in other organisms. The biosynthetic enzymes from these intact BGCs showed 53 to 71 percent sequence identity to the Cbp proteins, far higher than to other lasso peptide machinery in the same genome, supporting the functional assignment. The team also detected low-abundance compounds corresponding to mature lasso peptides from CbpA3 and CbpA4, confirming that the cbp machinery processes all four distantly encoded precursors.
The cebulassopins exhibit potent antiproliferative activity against human lung carcinoma cells, A549, with half-inhibitory concentrations of 0.79 and 1.47 micromolar for cebulassopins A and B, respectively. Cebulassopin A proved more potent than the clinical anticancer agent etoposide, which showed an IC50 of 2.57 micromolar. The compounds showed specificity, with only partial inhibition of glioblastoma cells and no effect on colon, breast, liver, or normal lung cells at concentrations up to 100 micromolar. Wound healing assays demonstrated that both compounds effectively block cancer cell migration without affecting cell morphology. This anticancer activity is unusual for lasso peptides, with only three prior examples reported. The split biosynthetic architecture opens new avenues for natural product discovery, as precursor-less BGCs may be more common than currently recognized and require chemistry-first approaches to uncover the products they generate.