For decades, a central axiom of cell-penetrating peptide, CPP, design has held that dense positive charge drives cellular entry. Linear CPPs such as Tat and R9 require at least six cationic residues, typically arginine or lysine, to cross the plasma membrane, and every additional charge carries a toxicity cost. Cyclization has helped, reducing the minimum requirement to four arginines in scaffolds like CPP12, but pushing below that threshold while preserving delivery efficiency has remained stubbornly difficult. The result is a charge-activity paradox: the same residues that power entry also poison cells.
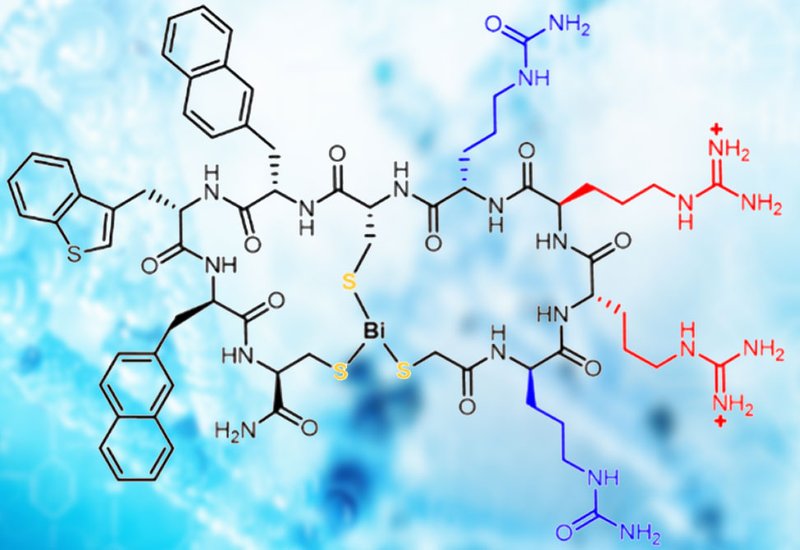
Researchers in the Pei Group at The Ohio State University, published in Biochemistry, conducted a systematic structure–activity relationship campaign on BCP16, a bismuth-mediated bicyclic CPP assembled via Bi3+ coordination of three thiol groups. Starting from the observation that BCP16 achieves excellent cytosolic delivery but carries cytotoxicity that limits its therapeutic potential, the team used medicinal chemistry optimization to interrogate every arginine position and redesign the scaffold's ring geometry, ultimately discovering BCP16e, an analog bearing only a +2 charge at physiological pH.
The team first established that Bi3+-binding affinity scales inversely with loop size: smaller rings preorganize the thiol ligands more favorably, requiring greater EDTA equivalents to strip the metal ion. Working from this insight, they replaced each of the four Arg or D-Arg residues of BCP16 individually with the neutral isostere citrulline, mapping which positions tolerated substitution without sacrificing entry. Positions one and four proved dispensable; positions two and three were critical. Replacing both dispensable arginines simultaneously, one D-citrulline at position one and one L-citrulline at position four, yielded BCP16e. The double substitution was not merely tolerated: BCP16e achieved 2.2-fold higher cytosolic entry efficiency than BCP16 in flow cytometry assays using naphthofluorescein-labeled peptides in HeLa cells, and its Bi3+-binding affinity rose 2.3-fold, likely because removing charged arginine side chains reduces repulsive interactions within the coordination sphere.
Confocal microscopy confirmed the quantitative picture. HeLa cells treated with 2 μM TMR-labeled BCP16e displayed intense, diffuse fluorescence throughout the cytoplasm and nucleus, a pattern consistent with efficient endosomal escape and cytosolic distribution, while CPP12 produced weaker, predominantly punctate signals. In a functional cargo-delivery test, BCP16e was conjugated to a peptidyl inhibitor of the Keap1–Nrf2 protein–protein interaction and evaluated using an antioxidant response element luciferase reporter assay in HepG2 cells. BCP16e-P1 achieved an EC50 of approximately 0.6 μM and induced up to approximately 70-fold luciferase activation, roughly twice as potent as the BCP16-P1 conjugate. Critically, BCP16e alone showed no cytotoxicity in HepG2 cells up to 10 μM, while BCP16 reduced cell viability by approximately 30% at the same concentration. In a dose-escalation intravenous study in BALB/c mice, BCP16 was lethal at 15 mg/kg, placing its LD50 between 10 and 15 mg/kg. Mice receiving BCP16e at 15 mg/kg, the highest dose tested due to solubility limits, showed only transient, reversible hypoactivity, with no mortality after seven days.
These findings directly challenge the assumption that high cationic charge is a prerequisite for membrane translocation, and they open a practical path for designing CPPs with improved therapeutic indices. Because BCP16e retains the same serum half-life as BCP16, t1/2 = 25 h in human serum, the safety gains come without sacrificing proteolytic stability. The authors note that the vesicle budding-and-collapse mechanism, which governs endosomal escape in this scaffold, can evidently be driven by hydrophobic aromatic residues as well as cationic ones, a mechanistic insight that should reshape how the field balances charge, hydrophobicity, and ring architecture in next-generation intracellular delivery vehicles.