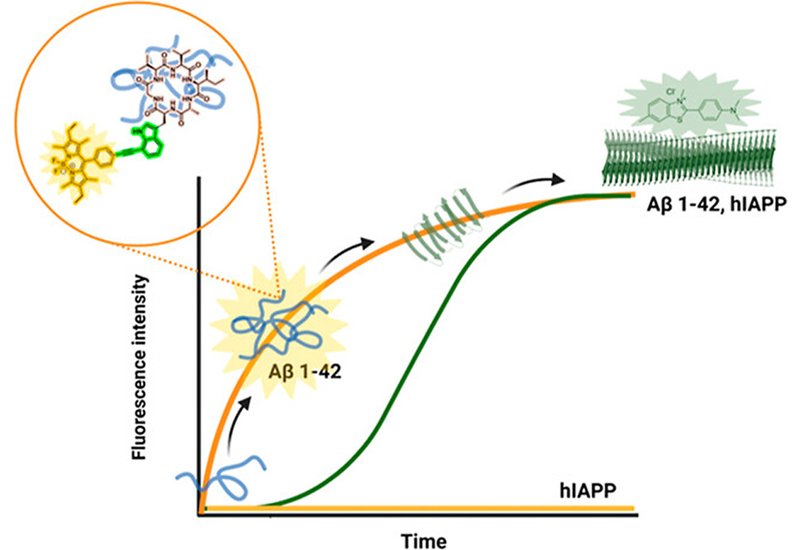
Early oligomeric aggregates of amyloid-β, Aβ1−42, are widely regarded as the most neurotoxic species in Alzheimer's disease, yet catching them in the act has proven stubbornly difficult. Their transient, structurally heterogeneous nature resists standard detection tools, and most fluorescent small-molecule probes that do respond to amyloid aggregation cannot distinguish Aβ from closely related peptides such as islet amyloid polypeptide, IAPP, the amyloidogenic culprit in type 2 diabetes. The field therefore needs probes that are simultaneously oligomer-selective and protein-selective, a dual specification that has not previously been achieved with a single molecular tool.
Researchers in the Tonali Lab at CEA Saclay, published in J. Med. Chem., addressed this challenge by combining rational cyclic peptide design with BODIPY fluorophore conjugation via late-stage palladium-catalyzed cross-coupling chemistry. The team drew on earlier work establishing that the C-terminal Aβ1−42 fragment spanning residues 38–42 can recognize Aβ oligomers; crucially, N-terminal acetylation of this fragment preserves oligomer binding while abolishing inhibitory activity, making it a clean scaffold for probe rather than therapeutic development. To boost conformational rigidity and proteolytic resilience, the sequence was embedded in a head-to-tail cyclic hexapeptide, c(-Val-Val-Ile-D-Ala-Trp-Gly-), where a single D-Ala residue enforces a well-defined double-β-turn architecture confirmed by NMR-restrained structure calculation in Amber with 23 distance restraints and 7 dihedral angle restraints. Three conjugation chemistries — Sonogashira cross-coupling, Mizoroki–Heck coupling, and copper-catalyzed azide–alkyne cycloaddition, CuAAC — were compared to evaluate how the linker geometry between the indole of bromotryptophan and the BODIPY core affected fluorogenic performance.
The Sonogashira conjugate, probe 8, carrying a rigid alkyne bridge between the tryptophan indole ring and the BODIPY phenyl core, delivered the sharpest fluorescence turn-on response. Under a standardized pH 7.4 aggregation protocol, probe 8 produced a 1.5-fold higher F/F0 value than the reference taBODIPY dye after 6 hours of Aβ1−42 incubation. At pH 8.0, where a defined lag phase isolates early prefibrillar species from mature fibrils, probe 8 reached maximum signal within 2 hours and showed a 1.7-fold advantage over taBODIPY at 6 hours, a window in which the classical fibril sensor Thioflavin T remains silent. Fluorescence titration experiments with monomeric Aβ1−42 yielded an apparent KD of approximately 70 nM for probe 8, while the Heck and CuAAC conjugates showed no measurable binding under the same conditions, underscoring the importance of the rigid, planar triple-bond linker for viscosity-sensitive fluorescence activation.
Selectivity experiments against IAPP revealed that probe 8 responded at only 0.2-fold the fluorescence level of taBODIPY during IAPP oligomerization kinetics, and titration experiments detected no measurable interaction with IAPP monomers. In contrast, the control dye lacking the cyclopeptide scaffold produced a strong, uncontrolled fluorescence increase upon IAPP oligomerization, confirming that the cyclic peptide module is the selectivity-determining element. In human SH-SY5Y neuroblastoma cells treated with monomeric Aβ1−42 for 24 hours, probe 8 revealed distinct intracellular puncta concentrated in the perinuclear region, a pattern consistent with aggregated Aβ species. Commercial anti-Aβ antibodies OC and D54 under identical conditions produced only diffuse cytoplasmic staining, demonstrating that probe 8 surpasses antibody-based immunofluorescence for visualizing intracellular amyloid aggregates.
This work establishes a modular platform in which a conformationally preorganized cyclopeptide confers molecular recognition specificity while a judiciously chosen conjugation chemistry tunes the fluorogenic response. The approach is generalizable: swapping the recognition sequence could, in principle, generate analogous probes for α-synuclein, tau, or IAPP aggregation. More immediately, probe 8 provides a practical tool for dissecting the earliest steps of Aβ aggregation in neuronal models and for screening inhibitors of prefibrillar species, a class of compounds that has proven difficult to evaluate with existing reporters.