Lasso peptides are among the most architecturally striking molecules in the natural product world. These ribosomally synthesized and post-translationally modified peptides range from 13 to 33 amino acids and adopt a topology in which the C-terminal tail threads through a macrolactam ring formed by an isopeptide bond between the N-terminal amine and the side chain of an aspartate or glutamate residue. Bulky residues above and below the ring act as steric plugs that lock the tail in place, conferring exceptional resistance to thermal denaturation and proteolytic degradation. More than 90 lasso peptides have been characterized to date, with genome mining predicting over 7,000 biosynthetic gene clusters encoding additional members. Their biological activities span antimicrobial, antiviral, anticancer, and enzyme-inhibitory functions, and their threaded structure is typically essential for potency: the unthreaded branched-cyclic isomer of microcin J25, for example, completely loses its ability to inhibit bacterial RNA polymerase.
In a new Perspective published in the Journal of the American Chemical Society, Professor David J. Craik and colleagues at the University of Queensland's Institute for Molecular Bioscience survey the rapidly evolving landscape of lasso peptide production. The biosynthetic pathway requires a minimum of three gene products: a precursor peptide, a leader peptidase, and an ATP-dependent lasso cyclase that installs the isopeptide bond around the pre-threaded tail. Recent structural and computational studies, including AlphaFold2-predicted complexes and molecular dynamics simulations, have begun to reveal how the recognition element, peptidase, and cyclase cooperate, with conserved hydrophobic residues in the leader peptide fitting into pockets on the recognition element in a knobs-into-holes arrangement. A single conserved threonine near the cleavage site has emerged as essential for proteolysis, and the loop region of the mature lasso peptide appears to be the primary contact surface for the cyclase.
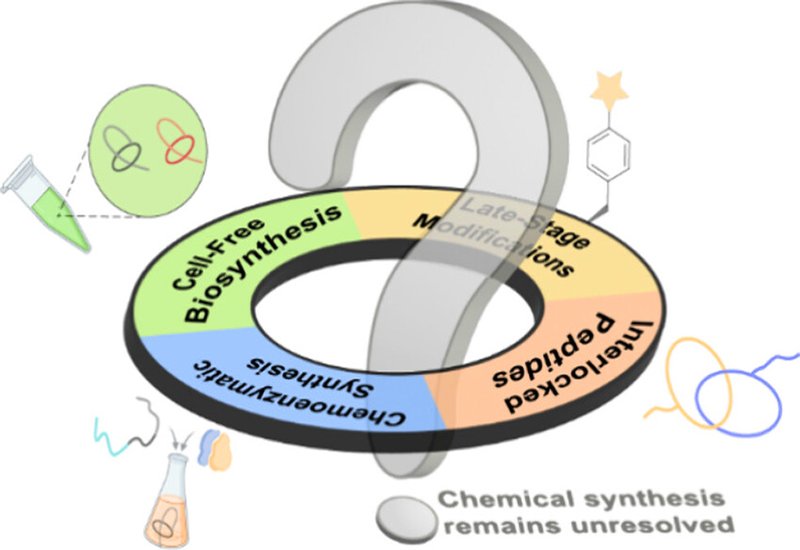
These mechanistic insights have fueled three production strategies that now complement traditional heterologous expression. Cell-free biosynthesis, introduced in 2021, achieves roughly 200-fold higher yields of capistruin compared to E. coli expression and enables rapid screening of precursor peptide variants. A chemoenzymatic approach combines chemically synthesized precursor peptides with enzymatic in vitro transformation, allowing incorporation of unnatural amino acids and backbone N-methylations that are inaccessible through genetic encoding alone. Late-stage chemical modification of recombinantly produced lasso peptides has introduced glycosylation, arylation, and non-native disulfide bonds at designed positions. On the applications front, engineered lasso peptides carrying grafted RGD epitopes have achieved nanomolar potency against cancer-associated integrins while retaining serum half-lives exceeding 30 hours, and lasso-inspired mechanically interlocked peptides have demonstrated thermal stability at 95 °C.
Despite this progress, a total chemical synthesis that achieves the native threaded fold remains elusive. Every purely synthetic attempt has produced only the unthreaded branched-cyclic conformation, underscoring how dependent the lasso topology is on enzyme-mediated pre-folding. The Perspective argues that computational methods, particularly molecular dynamics simulations capable of identifying synthetic strategies to stabilize the pre-lasso conformation, may ultimately bridge this gap. With the biological activities of more than 40% of known lasso peptides still uncharacterized, and post-translational modifications only beginning to be explored, the authors make a compelling case that lasso peptides represent one of the most promising and underdeveloped scaffolds in peptide-based drug design.