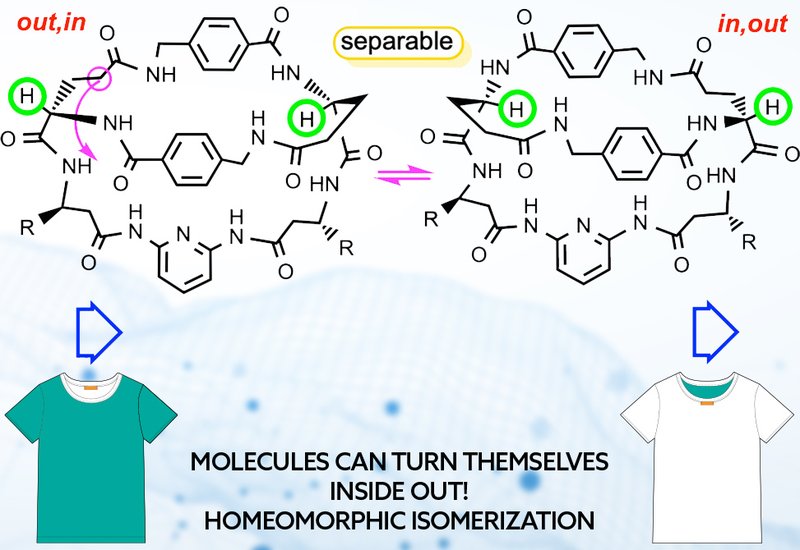
Macrobicyclic peptides have exploded onto the drug development scene over the past decade, attracting intense interest for oral drug delivery and therapeutic applications. Yet these molecules harbor a widely overlooked conformational phenomenon that could fundamentally alter their biological properties. When the tethers connecting the two bridgehead atoms are long and flexible enough, macrobicycles can undergo homeomorphic isomerization, a topological process that effectively turns the molecule inside-out. This transformation exchanges in and out bridgehead configurations without breaking any bonds, creating distinct structural forms, homeomorphs, with potentially different binding affinities, stabilities, and therapeutic activities.
Researchers in the Gladysz Group at Texas A&M University, published in the Journal of the American Chemical Society, have synthesized this critical but underappreciated concept for the peptide community. Drawing on decades of work with model compounds featuring phosphorus, arsenic, and other bridgehead atoms connected by methylene chains, the team demonstrates that homeomorphic isomerization occurs at room temperature when tethers contain ten or more sp³ carbon atoms. The process resembles pulling one tether through the ring formed by the other two, exchanging the molecular interior with the exterior. For macrobicyclic peptides with sufficiently long and flexible amino acid sequences connecting the bridgehead atoms, the same phenomenon applies. The team identified numerous peptide drug candidates, including compounds in clinical trials, where the specific homeomorph or bridgehead configuration remain undefined.
The implications span three time regimes. When homeomorphs interconvert slowly at room temperature, over hours or days, they can be separated and their individual properties exploited. Kilburn demonstrated this with a peptide-like macrobicycle where non-degenerate in,out and out,in isomers equilibrated with a half-life of 158 hours at 20 degrees Celsius. When the barrier is extremely high, homeomorphs represent distinct synthetic targets that would commonly be separable, as seen with amaninamide isomers that showed no interconversion even at 150 degrees Celsius in dimethyl sulfoxide, DMSO. When the barrier is very low, rapid equilibration creates opportunities for dynamic processes like metal ion capture and release, demonstrated by the team's dibridgehead diphosphines that transport platinum chloride through aqueous/organic interfaces. Crystal structures of alpha-amanitin, both free and bound to RNA polymerase II, revealed consistent out,out geometries with bridgehead carbon to bridgehead carbon to hydrogen angles between 129 and 155 degrees, though the possibility of alternative homeomorphs was never explored.
These findings create both opportunities and vulnerabilities. One intriguing application involves stealth drug delivery, where one homeomorph optimized for gastrointestinal survival is administered, then gradually isomerizes to the therapeutically active form at body temperature. The phenomenon also exposes gaps in intellectual property protection. Many macrobicyclic peptide patents fail to specify bridgehead configurations or consider homeomorphic forms, analogous to how older pharmaceutical patents overlooked enantiomeric composition. Compounds with bismuth or arsenic bridgeheads, which constitute stereogenic centers, are currently in intense development as therapeutics; these may be diastereomeric mixtures, and in no case have the absolute configurations been defined. The authors call for rigorous stereochemical characterization of all macrobicyclic peptides, assignment of in versus out bridgehead geometries using simple geometric criteria, and incorporation of homeomorphic considerations into computational drug design protocols that currently screen thousands of candidates without awareness of this conformational dimension.