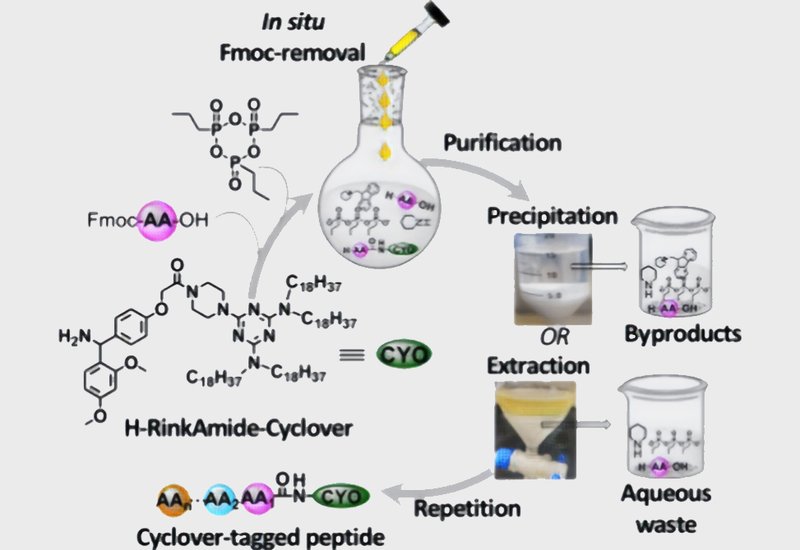
Peptide synthesis faces a persistent sustainability challenge. Solid-phase peptide synthesis, SPPS, dominates industrial production but requires large reagent excesses and generates substantial waste. Classical solution-phase synthesis offers high efficiency but demands labor-intensive chromatographic purification. Liquid-phase peptide synthesis, LPPS, has re-emerged as a middle ground, combining solution-phase efficiency with tag-based purification that avoids both chromatography and the extreme reagent excesses of SPPS. The growing peptide becomes soluble in nonpolar solvents via attachment to a solubility-modulating tag, allowing precipitation or extraction to replace filtration or chromatography. Yet most reported tags remain proprietary and commercially unavailable, limiting widespread adoption.
Researchers in the Albericio & de la Torre Group at the University of KwaZulu-Natal, published in Organic Letters, introduced Cyclover as a commercially accessible polycarbon tag for LPPS. Cyclover is a triazine scaffold bearing two long-chain dioctadecylamino groups and a piperazine handle for linker attachment. The team paired Cyclover with propylphosphonic anhydride, T3P, a coupling reagent recognized by the ACS Green Chemistry Institute Pharmaceutical Roundtable for its minimal environmental footprint. T3P generates water-soluble propylphosphonic acid as its sole byproduct, which aqueous extraction readily removes. The researchers performed all syntheses in 2-methyltetrahydrofuran, 2Me-THF, a greener solvent than dichloromethane or dimethylformamide. They synthesized Cyclover from cyanuric chloride in two steps, then coupled it to a RinkAmide linker using T3P and diisopropylethylamine, DIEA, in 2Me-THF. In situ Fmoc deprotection with piperidine followed directly in the same pot, eliminating the need for intermediate isolation. Neutralization with dilute hydrochloric acid and precipitation with acetonitrile afforded the deprotected handle in 98% yield.
The group demonstrated the method by synthesizing leucine-enkephalin, H-YGGFL-NH2, via iterative coupling, in situ deprotection, and isolation cycles. Each Fmoc-protected amino acid was coupled using 1.2 equivalents, T3P, and DIEA in 2Me-THF, followed by direct piperidine treatment for Fmoc removal. Two isolation strategies were compared: precipitation with acetonitrile and ethyl acetate versus aqueous extraction with dilute acid, sodium bicarbonate, and brine. Both routes delivered the pentapeptide in over 90% isolated yield after cleavage from the tag with trifluoroacetic acid and precipitation with tert-butyl methyl ether. High-performance liquid chromatography confirmed crude purities exceeding 95%, with the extraction route achieving over 98% purity. The team then synthesized protected linear oxytocin, a nine-residue sequence containing two acetamidomethyl-protected cysteines. Both precipitation and extraction routes afforded the protected nonapeptide in 86 to 87% isolated yield with crude purities above 95%. Phosphorus-31 nuclear magnetic resonance confirmed complete removal of T3P-derived byproducts in all final products.
Green metrics revealed a striking advantage for the extraction protocol. Process mass intensity, PMI, and complete environmental factor, cEF, quantify total mass input per unit product, including all solvents and water. For leucine-enkephalin, precipitation yielded PMI and cEF values of 8426 and 8425, whereas extraction reduced these to 3182 and 3181, a 2.7-fold improvement. The reduction arose from decreased solvent volumes in extraction relative to the large excesses required for precipitation at each cycle. Although these values remain higher than those for optimized SPPS with in situ deprotection, PMI 571 and cEF 570, the unoptimized LPPS method surpasses standard SPPS, PMI 2243 and cEF 2242. The water-soluble nature of T3P byproducts proved critical, enabling simple aqueous washes to replace chromatography or complex precipitation protocols. Cyclover's tunable solubility in 2Me-THF for coupling and insolubility in polar solvents for precipitation simplified workup across both isolation strategies. The use of 2Me-THF, a bio-derived solvent with favorable environmental profiles, further enhanced the method's green credentials. Dichloromethane synthesis required less T3P, 6 equivalents versus 8, due to lower moisture uptake, but 2Me-THF delivered comparable purity while avoiding chlorinated solvents.