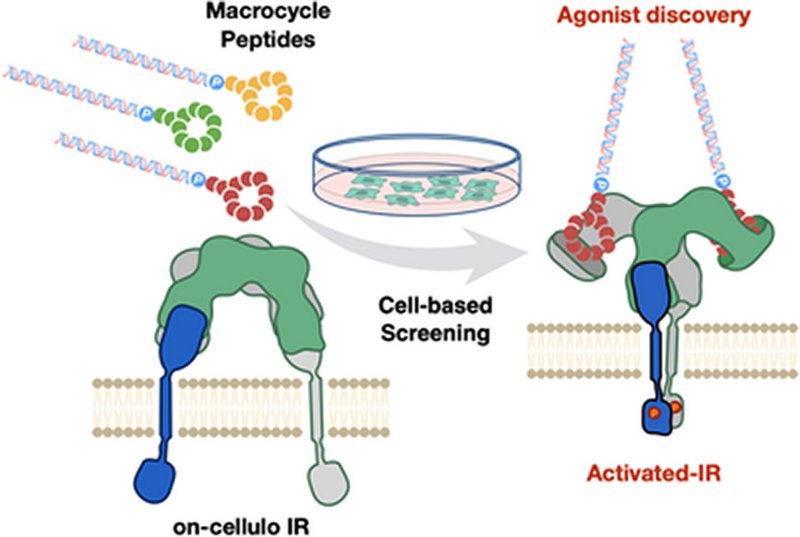
Macrocyclic peptides have emerged as a compelling therapeutic modality for difficult protein targets, and mRNA display technologies now allow researchers to screen libraries exceeding one trillion unique sequences. Yet a persistent gap separates the discovery of tight binders from the discovery of functional agonists. Affinity-based selection against purified receptor fragments cannot guarantee that enriched sequences will engage a receptor in its native membrane context, preserve the oligomeric architecture needed for signaling, or trigger the conformational changes that activate a kinase domain. For the insulin receptor, IR, a tyrosine kinase whose dysregulation drives diabetes and cancer, this gap has kept de novo macrocyclic agonists out of reach.
Researchers in the Suga Group at the University of Tokyo, published in Angewandte Chemie International Edition, designed an upgraded selection platform called RaPID-ExCells, RaPID Extended to Cell-based screening, that integrates the genetic code reprogramming and mRNA display power of the established RaPID system with direct functional screening against receptors on living cells. Rather than selecting against an immobilized ectodomain fragment, the team exposed enriched macrocycle libraries to intact HEK293 cells, lysed the cells with a nonionic detergent that preserves native IR conformation and dimerization, and recovered peptide–mRNA/cDNA conjugates bound to endogenous IR via co-immunoprecipitation with an anti-IR monoclonal antibody. Over three rounds of ExCells selection, the positive-to-negative recovery ratio climbed dramatically, signaling robust enrichment of on-cellulo binders.
Sequencing the enriched populations by next-generation sequencing and visualizing them with UMAP revealed that the ExCells pressure collapsed a broad field of ectodomain binders into just four clusters, with one sequence, HL4, reaching roughly 50% of the total population by round five. When the team synthesized the top eleven affinity-selected peptides and tested them by western blot for IR autophosphorylation, only HL4 activated the receptor. Crucially, surface plasmon resonance showed that HL4 bound the isolated IR ectodomain with a KD of 132 nM, weaker than several competing sequences with KD values in the 2–50 nM range. The result made the point unmistakably: affinity alone does not predict agonist function, and a function-first selection was required to find the active molecule.
To probe the structural determinants of HL4 activity and push its potency further, the researchers conducted deep mutational scanning, DMS, using RaPID-ExCells itself as the readout. Scanning 66 amino acids, including 47 non-proteinogenic amino acids, npAAs, across each position revealed that the FYLWF aromatic motif at residues 9–13 is essential for receptor activation, mirroring the FYXWF pharmacophore previously identified in linear insulin-mimetic peptides from phage display. Substitutions at position F9 with moderately bulky para-substituted phenylalanine analogs, including p-methyl and p-fluoro variants, enhanced agonist activity roughly 1.5-fold. Glycine at position 5 tolerated D-amino acid substitutions, consistent with its proposed role as a turn-inducing residue, and D-Tyr at this site modestly boosted activity while offering a handle for improved proteolytic stability. A homodimeric HL4 construct, two monomers linked through PEG11 spacers to an L-Dap core, achieved an EC50 of 460 nM, showed 2.5-fold higher autophosphorylation than the monomer at 1 μM, extended the serum half-life to 51 hours, and activated IR selectively without triggering related receptor tyrosine kinases including IGF-1R.
RaPID-ExCells reframes the logic of macrocycle discovery by making biological function, rather than isolated affinity, the primary selection criterion. The platform is not limited to the insulin receptor and should generalize to any cell-surface receptor or membrane protein that can be captured by co-immunoprecipitation after mild lysis. By folding DMS of npAAs directly into the same cell-based readout, the approach compresses what would otherwise be iterative cycles of synthesis, assay, and redesign into a single high-throughput campaign. The work positions RaPID-ExCells as a broadly applicable strategy for finding macrocyclic leads that engage clinically relevant receptors in the context that actually matters, the living cell surface.