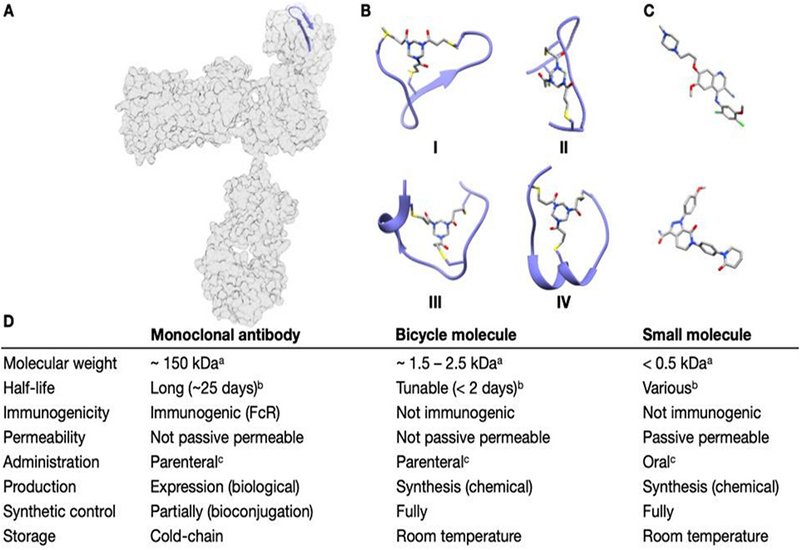
Bicyclic peptides gain increasing attention in drug development given their unique properties which combine the synthetic customizability of small molecules with the precision of antibodies, yet with differentiated pharmacokinetic profile, avoiding Fc-related pharmacology while enabling deep tissue penetration and tunable clearance. Methods for generating these constrained structures, which made it into clinical development, rely on C3-symmetric organic scaffolds that link covalently three cysteines in a peptide chain, thus locking the peptide into a conformation that improves target affinity and elongates proteolytic stability. This approach has powered clinical-stage programs such as those at Bicycle Therapeutics and generated a large portfolio of preclinical candidates against a wide range of protein targets.
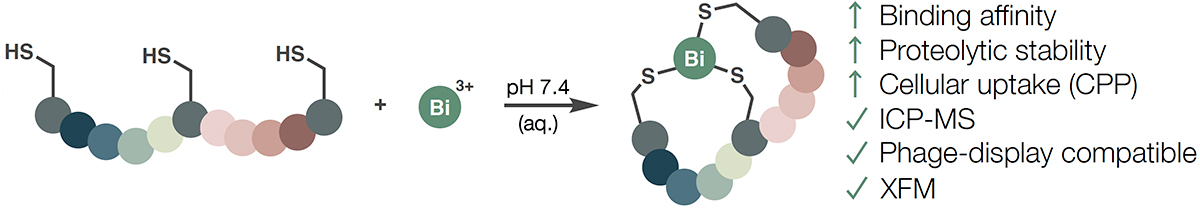
Researchers in the Spring Group at the University of Cambridge, working in collaboration with Bicycle Therapeutics, reflect on the discovery of bismuth(III) as an alternative scaffold for bicyclic peptide synthesis, published in the Journal of Peptide Science. Rather than relying on the electrophilicity of organic linkers, this method leverages the thiophilic nature of bismuth: Bi3+ forms selective, stable complexes with three cysteine thiolates rapidly, producing a bicyclic architecture anchored by a single metal atom. At an atomic radius of just 1.5 Å, bismuth represents the smallest scaffold yet applied to bicyclic peptide synthesis, generating complexes that are more constrained than those produced by TBMB, TATA, TBAB, or TATB, as the researchers demonstrate through computational analysis. Cyclisation proceeds instantaneously at physiological pH, tolerates the reducing agent TCEP, and requires no organic solvent, placing bismuth bicyclisation among the mildest and most operationally straightforward methods available.

From left to right: Dr. Amin Sagar, Assoc. Director, Computational Science, Dr. Inma Rioja, Senior Vice President, Head of Therapeutics, Dr. Mike Skynner, Chief Scientific Officer, Dr. Saan Voss, UKRI Knowledge Transfer Partner, Dr. Liuhong Chen, Vice President, Phage Innovation Platform, and Dr. Mark Frigerio, Vice President, Head of Platform Technology & Science.
Bicycle Therapeutics is one of the leading constrained peptide companies whose technology has been driving innovation in a number of areas including the targeted radiopharmaceuticals space. The team led by Dr Mike Skynner, Chief Scientific Officer at Bicycle Therapeutics, is developing a portfolio of Bicycle Drug Conjugate therapeutics and has pioneered Bicycle® binders against MT1-MMP and EphA2, two previously considered undruggable targets, which have undergone first-in-human imaging.
More recently Bicycle signed a first of its kind deal with the UK Nuclear Decommissioning Authority and UK National Nuclear Laboratory to turn reprocessed uranium, a byproduct of nuclear power plants, into lead-212 for precision guided cancer therapies using a 212Pb generator being developed exclusively for Bicycle by SpectronRx. The team does not stop there and continues to reimagine molecular architectures to innovate the next generation of molecules, such as Bismuth Bicycles.
The structural constraint imposed by bismuth translates into measurable gains across multiple biological dimensions. Bismuth Bicycle molecules inhibited proteases from Zika and West Nile viruses with greater potency than their linear counterparts, exceeding the activity of the linear congener against West Nile virus protease by more than two orders of magnitude. Proteolytic stability improved 6- to 19-fold depending on sequence and protease identity. In cell-penetrating peptide variants, bismuth cyclisation enhanced cellular uptake by 10-fold relative to the linear counterpart and exceeded benchmark CPPs such as Tat49-57 and R8, with cellular uptake detectable across multiple cancer cell lines and in some cases at concentrations as low as 10 nM. The heavy metal core also enables detection by inductively coupled plasma-mass spectrometry (ICP-MS) and X-ray fluorescence microscopy (XFM), analytical approaches that bypass fluorescent labelling and permit quantification of unlabeled compounds directly from biological matrices. Furthermore, XFM also enables the subcellular localization of Bismuth Bicycle molecules for instance to study a cargo's effect on cellular update. Bi3+ salts are also compatible with phage display, enabling genetically encoded bismuth Bicycle libraries from which binders against model proteins were enriched with dissociation constants two orders of magnitude tighter than those observed without bismuth.
The convergence of bismuth's biological tolerability, its utility as a scaffold, and its potential as a radioisotope positions this compound class at an unexplored intersection of inorganic chemistry and chemical biology. For instance, Bismuth-213, a radioactive isotope with a half-life of approximately 46 minutes, undergoes alpha decay and could replace stable bismuth-209 in Bicycle molecules to yield precision radiopharmaceuticals for targeted alpha therapy in oncology. Combined with phage display selection against clinically relevant targets, bismuth Bicycle molecules may offer a new route to diagnostics and therapeutics in cancer and infectious disease.